Patient Information
Welcome to our Liver Cancer Information Hub. If you or a loved one is facing a diagnosis of liver cancer, you’re not alone. This website is designed with you in mind, providing clear and reliable information about liver cancer, its various forms, treatment options, and ways to navigate this challenging journey.
Our goal is to empower you with knowledge, so you can make informed decisions about your healthcare and better understand what to expect throughout the process. From understanding the basics of liver cancer to exploring the latest treatment advancements, we’re here to support you every step of the way.
What is liver cancer?
Liver cancer is defined by an abnormal and uncontrolled growth of cells within the liver. These altered cells progressively substitute the normal cells, collapsing the normal function of the liver, and invading other organs.
We should distinguish liver cancer from benign tumours that are also the result of altered cellular growth, but they lack the invasiveness capacities of malignant tumours (cancer). Therefore, the prognosis of benign tumours is excellent, and, in most cases, there is no need to treat them.
Briefly, there are two main types of liver cancer: primary liver cancer, which appears from the cells that are normally located in the liver, and secondary liver cancer (also known as metastases), which is cancer generated in other parts of the body (in the lungs, breasts or in the colon for instance) that sends the diseased cells to the liver, where these are implanted. The treatment of this secondary liver cancer depends on the type of primary cancer from which it has originated and is mainly based on the use of pharmaceutical drugs, commonly known as chemotherapy.
This information focuses on primary liver cancer. There are several types of primary liver cancer according to the type of cells that are altered. The most frequent, by far, is hepatocellular carcinoma (commonly referred to as HCC), which is a cancer derived from the main cells of the liver called hepatocytes. There are other types of primary liver cancer such as intrahepatic cholangiocarcinoma (iCCA, derived from the biliary cells), sarcomas, etc. This should also be taken into account.
Helpful resources
The International Liver Cancer Movement (ILCM) provides useful resources that raise awareness and explain liver cancer, it’s risk factors and possible treatments.

Learn more about Liver Cancer to be aware of it’s potential risk and treatments through short educational videos on ILCM website.

As early-detection is crucial, you can complete a self-assessment checkbased on the main risk factors for liver cancer (HCC and CCA) developed by ILCM.
Learn more about Liver Cancer
What is hepatocellularcarcinoma?
Hepatocellular carcinoma is a cancer that develops from the liver’s hepatocytes. Hepatocellular carcinoma is the most common form of liver cancer. It ranks as the fifth most common cancer worldwide. It is the third leading cause of cancer-related deaths, responsible for 500,000 to 1 million deaths annually. Its incidence has significantly increased since the mid-2000s. It primarily affects individuals over 50 years of age and is more common in men than women. Risk factors for Hepatocellular carcinoma include liver cirrhosis (scarring of the liver), infections from hepatitis B and C viruses, presence of metabolic syndrome and diabetes, exposure to certain toxic chemicals, and excessive alcohol consumption.
For patients with Hepatocellular carcinoma risk factors, enhanced monitoring of biological and morphological parameters is now established to detect the disease at an earlier stage. This involves performing abdominal ultrasound and measuring alpha-fetoprotein (AFP) levels every 6 months. Diagnosis of Hepatocellular carcinoma relies on biopsy (a procedure where cells or a small piece of tissue is obtained from the body) or imaging techniques meeting specific criteria. Some clinical signs may appear in the advanced stages of the disease, including abdominal pain, jaundice, weight loss, and fatigue.
Treatment choices depend on tumour size, location, the liver’s function, and the presence of metastases. Like other types of cancer, treatment decisions are made during a multidisciplinary tumour board meeting involving specialists in liver tumour management (surgeons, hepatologists, oncologists, radiologists, radiotherapists, etc.). A personalized care plan is then discussed with the patient in a dedicated consultation.
Curative treatments for Hepatocellular carcinoma include liver transplantation, surgical resection, and ablation techniques, all aimed at curing the cancer whenever possible. When curative treatments are not feasible, options include chemoembolization, external radiotherapy, selective internal radiotherapy (radioembolization), combination immunotherapy + anti-angiogenic agents, or tyrosine kinase inhibitors.
The prognosis is less favourable in patients with advanced-stage Hepatocellular carcinoma, but positive advancements in the field offer hope for the future. Advanced-stage Hepatocellular carcinoma cases that were previously inoperable can now be treated due to these interventions. The prognosis for Hepatocellular carcinoma patients depends on numerous factors, including disease stage, concurrent illnesses, and treatment response.
In conclusion, Hepatocellular carcinoma is the most prevalent form of liver cancer, often diagnosed at an advanced stage with a bad prognosis. The development of new therapeutic techniques and ongoing clinical trials aim to improve patient prognosis.
Reference: Vogel A, Meyer T, Sapisochin G, Salem R, Saborowski A. Hepatocellular carcinoma. Lancet. 2022 Oct 15;400(10360):1345-1362. doi: 10.1016/S0140-6736(22)01200-4. Epub 2022 Sep 6. PMID: 36084663.
What is cholangiocarcinoma?
Cholangiocarcinoma is a rare form of cancer that originates from the cells of the bile ducts within the liver. Cholangiocarcinoma can affect the intrahepatic bile ducts (inside the liver) as well as the main bile duct. It primarily affects individuals over 50 years of age and is more common in men than women. Risk factors for cholangiocarcinoma include liver cirrhosis, primary sclerosing cholangitis (inflammation and scarring of the bile ducts in the liver), infections from hepatitis B and C viruses, presence of metabolic syndrome and diabetes, parasitic infection by liver flukes, intrahepatic gallstones, choledochal cysts, exposure to certain toxic chemicals, and excessive alcohol consumption.
Diagnosing cholangiocarcinoma is often challenging as patients do not typically have specific symptoms in the early stages of the disease. Clinical signs may emerge at a more advanced stage and can include abdominal pain, jaundice (yellowing of the skin, whites of the eyes and mucous membranes), weight loss, and fatigue. When cholangiocarcinoma is suspected, a series of biological tests and imaging assessments (CT scan, MRI) will be conducted.
In cases of jaundice indicating bile duct obstruction, endoscopic stent placement may be considered to alleviate the obstruction and allow bile flow. The diagnosis of cholangiocarcinoma is confirmed through a biopsy, either transhepatic or endoscopic, depending on the location of the tumour within the bile ducts. These tests help determine the disease stage and guide treatment decisions.
The treatment approach depends on tumour size, location, liver function, and the presence of metastases. Similar to other types of cancer, treatment decisions are made during a multidisciplinary tumour board meeting involving specialists in liver tumour management. A personalized care plan is then presented to the patient during a dedicated consultation.
Surgery is often the preferred treatment for localized tumours, although its feasibility depends on the proximity of the tumor to the bile ducts and blood vessels. Approximately 25% of diagnosed cholangiocarcinomas are eligible for surgical intervention. When surgery is not possible, localized treatments such as ablation, radioembolization, or external radiotherapy may be considered. Liver transplantation for cholangiocarcinoma is rare due to a high risk of recurrence post-transplantation and is a subject of ongoing research.
For advanced-stage disease, patients who cannot undergo localized hepatic treatment or present with metastases are candidates for systemic treatments like chemotherapy, immunotherapy, or targeted therapies. Currently, the first-line treatment combines intravenous chemotherapy (Gemcitabine-Cisplatin) with immunotherapy (Durvalumab).
The prognosis for cholangiocarcinoma patients depends on numerous factors, including disease stage, concurrent illnesses, and treatment response. Unfortunately, the five-year survival rate for cholangiocarcinoma patients is generally low, with an overall median survival of 12 months for all types of cholangiocarcinomas combined.
In conclusion, cholangiocarcinoma is a rare form of cancer often diagnosed at an advanced stage with a grim prognosis. The development of new therapeutic techniques and ongoing clinical trials aim to improve patient prognosis.
Reference: Brindley, P.J., Bachini, M., Ilyas, S.I. et al. Cholangiocarcinoma. Nat Rev Dis Primers 7, 65 (2021). https://doi.org/10.1038/s41572-021-00300-2
What is liver transplantation and in which situation can this treatment be proposed in the setting of liver cancer?
Liver transplantation for cholangiocarcinoma is rare due to an elevated risk of recurrence post-transplantation and is a subject of ongoing research.
Some patients with hepatocellular carcinoma can benefit from treatment through liver transplantation. The decision for this treatment is made during a multidisciplinary tumour board meeting involving all specialists involved in liver tumour management (surgeons, hepatologists, oncologists, radiologists, radiotherapists, etc.) and depends on factors such as tumour size, location, presence of metastases, and the function of the unaffected liver. Liver transplantation treatment is typically reserved for patients with early-stage hepatocellular carcinoma who are not suitable candidates for surgical or ablative treatments, or in cases of hepatocellular carcinoma recurrence after such treatments.
The goal of liver transplantation is to replace the diseased liver with a healthy liver from a compatible donor, thereby eliminating the tumor and underlying liver disease. The procedure can take several hours and requires an extended hospital stay for careful monitoring. Side effects associated with liver transplantation may include post-operative pain, fatigue, nausea, and loss of appetite. Patients also need to take immunosuppressive medications (medications that suppress an immune response) to prevent rejection of the new liver, which can lead to long-term complications such as infections, cancer, or kidney failure.
Patient selection for liver transplantation is based on various tumour-related factors, including tumour size, number of tumours, and AFP levels, as well as age and presence of comorbidities (presence of other diseases). Only patients with active early-stage hepatocellular carcinoma are eligible for this type of treatment due to the significant risk of recurrence under immunosuppression. In cases of advanced tumours, downstaging may be necessary. This involves reducing the tumour size with prior treatments such as radioembolization, chemoembolization, or radiotherapy to enable subsequent liver transplantation.
Surveillance is a crucial aspect of liver transplantation. Patients need to undergo regular examinations (laboratory tests and imaging) to ensure the new liver is functioning properly and there are no signs of hepatocellular carcinoma recurrence.
In conclusion, liver transplantation is an effective treatment option for patients with hepatocellular carcinoma under the age of 70, who have severe liver disease and a tumour that cannot be surgically removed or treated with ablation, or in cases of recurrence after these treatments.
Is surgery indicated for liver cancer?
Patients with hepatocellular carcinoma or cholangiocarcinoma can benefit from surgical treatment. The decision for this treatment is made during a multidisciplinary tumour board meeting involving all specialists involved in liver tumour management (surgeons, hepatologists, oncologists, radiologists, radiotherapists, etc.) and depends on factors such as tumour size, location, presence of metastases, and the function of the unaffected liver. Surgical treatment is generally reserved for single or multiple tumours located within the same liver segment and for patients with preserved liver function without clinically significant portal hypertension. The treatment and personalized care plan will be presented to the patient following the tumour board meeting during a dedicated consultation.
The primary goal of surgical treatment is to completely remove the cancerous tumour while preserving the remaining liver function. New techniques, including laparoscopy, enable less invasive surgical interventions, leading to quicker recovery and reduced postoperative complications. Laparoscopy is a minimally invasive surgical technique that utilizes thin instruments and a small tube equipped with a camera to visualize the inside of the body. This technique allows for more precise and less invasive surgery for patients with hepatocellular carcinoma.
Patient selection for surgery depends on factors such as underlying liver function, remaining liver volume, and the presence or absence of portal hypertension. Patients need to undergo a series of assessments before the intervention to evaluate their health status and suitability for surgery based on these criteria.
In certain cases, if the remaining liver volume is too small for surgical intervention, a technique called portal embolization can be used as an adjunct treatment. This technique involves blocking the vein that supplies the liver to induce atrophy of the cancerous liver and allow for regeneration and an increase in the volume of the non-tumour liver, with the goal of performing surgery at a later stage.
Side effects of surgery may include postoperative pain, fatigue, loss of appetite, nausea, vomiting, and decompensation of cirrhosis.
Post-treatment surveillance is important to detect any recurrence or reappearance of the tumour. Patients will be regularly monitored by their primary physician, and imaging examinations such as CT scans or MRIs will be performed every 3 months to monitor disease progression.
In conclusion, surgery is an effective treatment option for patients with hepatocellular carcinoma with one or multiple lesions located within the same liver segment. It is reserved for patients with preserved liver function and in the absence of significant portal hypertension. New techniques such as laparoscopy allow for less invasive surgery with faster recovery and reduced postoperative complications.
What refers to ablative therapies in the setting of liver cancer?
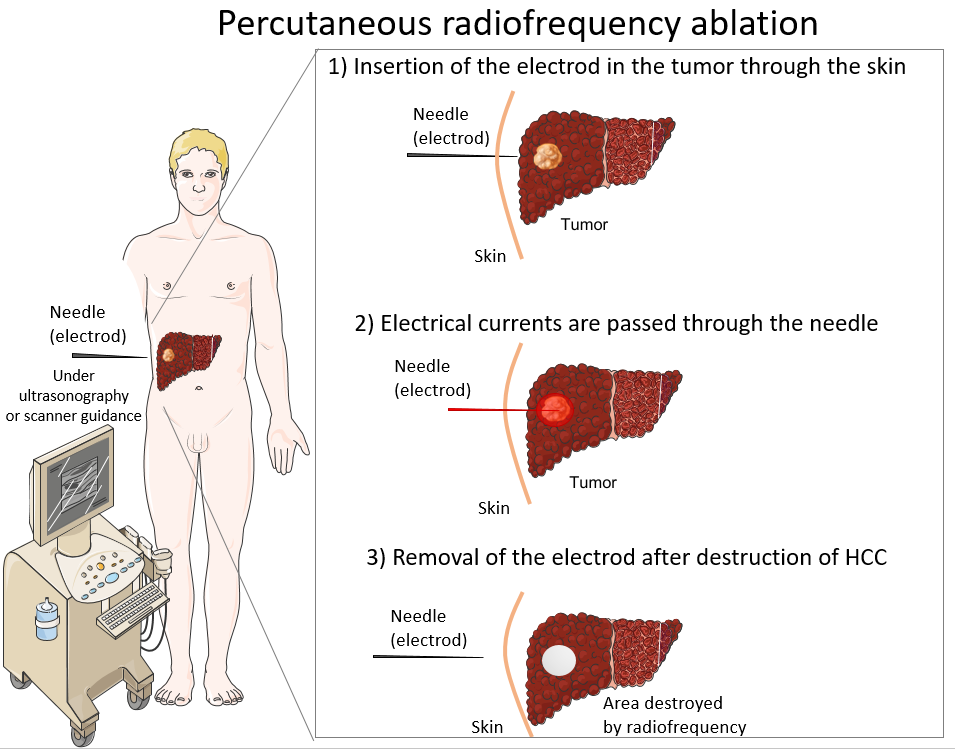
Percutaneous ablation treatment is a common method used to treat hepatocellular carcinomas (HCC). This technique can also be employed for the treatment of small-sized cholangiocarcinomas that are not accessible to surgical treatment. Needles are inserted through the skin to deliver heat or cold to destroy cancer cells. Several percutaneous ablation techniques exist for treating hepatocellular carcinomas, including radiofrequency, cryoablation, and microwave thermoablation. The most commonly used technique is percutaneous radiofrequency ablation.
Percutaneous ablation treatment requires selecting patients who can benefit from this type of approach. Before treatment, a comprehensive preoperative assessment must be conducted to determine the size, number, and location of the tumour (s). This assessment is typically performed using medical imaging, such as ultrasound, computed tomography (CT), or magnetic resonance imaging (MRI). The decision for this treatment is made during a multidisciplinary tumour board meeting involving all specialists involved in liver tumour management (hepatologists, oncologists, radiologists, radiotherapists, surgeons, etc.), and the possibility of undergoing this treatment depends on factors such as tumour size, location, presence of metastases, and the function of the unaffected liver. The treatment and personalized care plan will be presented to the patient following the multidisciplinary tumour board meeting during a dedicated consultation.
Percutaneous ablation is performed under general anaesthesia. Percutaneous radiofrequency ablation and microwave ablation are the most used types of percutaneous ablation. One or more needles are inserted through the skin and guided to the tumour using medical imaging. Once in place, the needle’s tip is heated to a high temperature to destroy cancer cells. Note that certain percutaneous ablation methods such as irreversible electroporation or cryoablation do not use heat as a method of destroying cancer cells. After treatment, patients must be closely monitored for 24 to 48 hours to detect side effects and manage potential complications. Percutaneous ablation does not involve major surgery or incisions and is associated with a low risk of complications.
Imaging examinations are performed regularly to ensure that the tumour has been completely destroyed and to detect any potential cancer recurrence. The advantages of percutaneous ablation treatment include low procedure-related morbidity and mortality, short hospitalization, and the possibility of repeating the treatment in case of localized and small-sized cancer recurrence within the liver.
In conclusion, percutaneous ablation treatment is an effective method for treating hepatocellular carcinomas in patients and is currently considered one of the main curative treatments for hepatocellular carcinoma.
When can chemoembolization be proposed to patients with liver cancer?
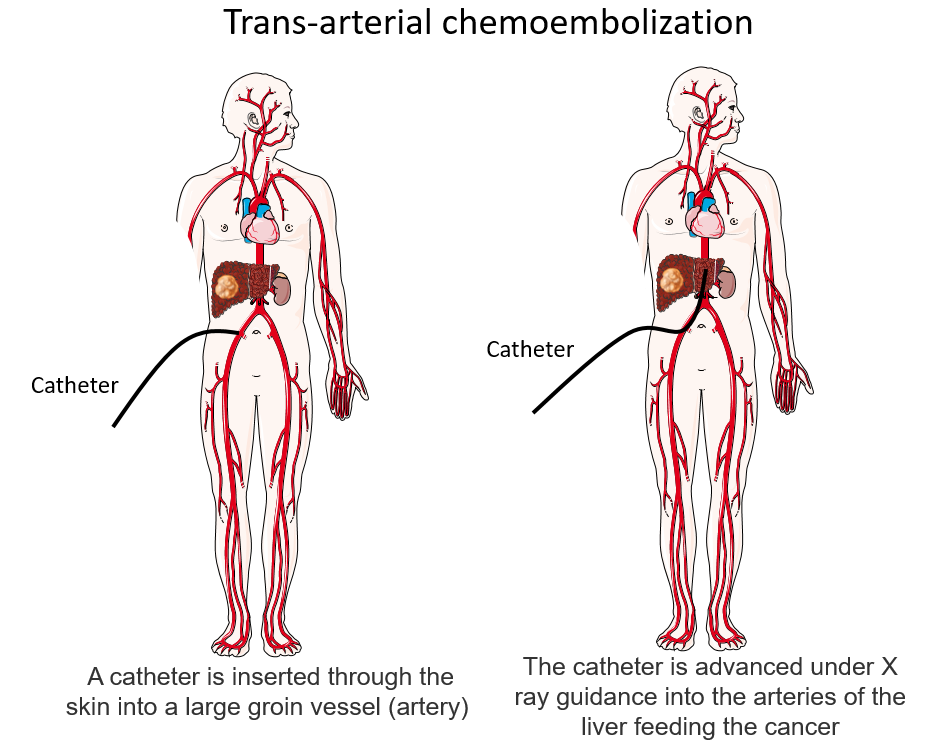
Patients with hepatocellular carcinoma or neuroendocrine tumours can benefit from treatment through chemoembolization. The decision for this treatment is made during a multidisciplinary tumour board meeting involving all specialists involved in liver tumour management (surgeons, hepatologists, oncologists, radiologists, radiotherapists, etc.) and depends on factors such as tumour size, location, presence of metastases, and the function of the unaffected liver. Chemoembolization treatment is generally reserved for multiple tumours that are not accessible to surgical or ablative treatment and for patients with preserved liver function. This treatment can also serve as a waiting option before surgery or liver transplantation. The treatment and personalized care plan will be presented to the patient following the tumour board meeting during a dedicated consultation.
Chemoembolization is an interventional radiology technique performed by a radiologist and consists of two steps:
- A fatty substance mixed with chemotherapy drugs is injected into arteries near the tumor to destroy liver tumor cells through a cytotoxic effect.
- Blood flow within the tumour vessels is significantly reduced or eliminated using embolization agents.
This technique allows for targeted action on tumours while preserving healthy liver tissue.
General anaesthesia is not required for this type of procedure, and chemoembolization treatment is carried out as a short hospitalization, typically lasting 2-3 days. The procedure begins with puncturing the radial or femoral artery, followed by catheterization to access the hepatic artery and inject the chemical and particle mixture into the tumour. Depending on the number and location of lesions, multiple successive procedures spaced 4-6 weeks apart may be necessary to control the disease.
In the days following chemoembolization, it’s not uncommon for patients to experience a set of symptoms known as post-chemoembolization syndrome, including abdominal pain, nausea and/or vomiting, fever, and hair loss. These side effects are generally temporary and can be managed with medications or symptomatic treatments.
Note that this technique is not indicated for patients with:
- Impaired liver function.
- Renal insufficiency.
- Extrahepatic metastases.
- Severe heart failure.
Post-treatment surveillance is important to evaluate the response to this therapy. Patients will be regularly monitored by their primary physician, and imaging examinations such as CT scans or MRIs will be performed every 3 months to monitor disease progression.
In conclusion, chemoembolization is an effective treatment option for patients with hepatocellular carcinoma presenting multiple intrahepatic tumours that are not accessible to surgery or ablation. Its aim is to reduce tumor size and prevent its growth.
What corresponds to a radioembolization treatment?
Liver tumours such as hepatocellular carcinoma and cholangiocarcinoma can be treated with radioembolization, also known as selective internal radiation therapy. The decision for this treatment is made during a multidisciplinary tumour board meeting involving all specialists involved in liver tumour management (surgeons, hepatologists, oncologists, radiologists, radiotherapists, etc.), and the possibility of undergoing this treatment depends on factors such as tumour size, location, presence of metastases, and the function of the unaffected liver. Radioembolization treatment is generally reserved for single tumours that are not accessible to surgery, ablation, or chemoembolization, and for patients with preserved liver function. This treatment can also be considered as a waiting treatment before surgery or liver transplantation. The treatment and personalized care plan will be presented to the patient following the multidisciplinary tumour board meeting during a dedicated consultation.
Radioembolization is an anticancer treatment aimed at slowing down or reducing the size of the tumour. For this purpose, millions of microscopic radioactive beads (SIR-Spheres or TheraSpheres measuring about 25 microns) are injected into the arteries that supply the tumour. These beads travel through the blood vessels that irrigate the tumour and destroy the cancer cells. The radiation emitted by the yttrium-90 present on the surface of each sphere only affects a few millimetres. Therefore, the surrounding healthy tissues are generally spared.
Radioembolization is an interventional radiology technique performed by a radiologist in two steps: The first step is the preparatory phase. In interventional radiology, a catheter is introduced into the femoral artery located at the root of the thigh, or through the radial artery located at the wrist, and then directed towards the hepatic artery. Once the catheter is positioned near the tumour, the radiologist checks the tumour’s blood supply and injects the radioactive micro-aggregate albumin preparation into the tumour. The non-toxic product is used to verify the proper fixation of the product at the tumour site and the absence of fixation outside the tumour in the liver and elsewhere in the body. The patient is then transferred to nuclear medicine for a scintigraphy scan. This examination is painless.
The second step is the therapeutic phase, which is similar to the preparatory phase except for the injection of radioactive beads. This procedure is performed under local anaesthesia.
Note that this technique is not indicated for patients with:
Unpreserved liver function;
Renal insufficiency;
Extrahepatic metastases;
Major heart failure;
Radioembolization can be performed during a short hospitalization, usually for 2-3 days per treatment phase, or on an outpatient basis depending on the centres. The procedure may be associated with side effects such as nausea, fatigue, and mild abdominal pain. These side effects are generally temporary and can be managed with medications or symptomatic treatments.
Post-treatment surveillance is important to evaluate the treatment response. Patients will be regularly followed by their referring physician, and imaging examinations such as CT scans or MRIs will be conducted every 3 months to monitor the disease’s progression. The true effectiveness of the treatment is typically assessed around 6 months after treatment due to the prolonged action of radiation.
In conclusion, radioembolization is an effective treatment option for patients with hepatocellular carcinoma that is not accessible by surgery, ablation, or chemoembolization. It allows for directly targeting cancer cells in the liver with minimal side effects.
Is there some indication for external radiotherapy in the setting of liver cancer?
Liver tumors such as hepatocellular carcinoma and cholangiocarcinoma can be treated with external beam radiotherapy. The decision for this treatment is made during a multidisciplinary tumor board meeting involving all specialists involved in liver tumor management (surgeons, hepatologists, oncologists, radiologists, radiation therapists, etc.), and the possibility of undergoing this treatment depends on factors such as tumor size, location, presence of metastases, and the function of the unaffected liver. External beam radiotherapy treatment is generally reserved for single tumors that are not accessible to surgery, ablation, or intra-arterial treatments (chemoembolization, radioembolization), and for patients with preserved liver function. The treatment and personalized care plan will be presented to the patient following the multidisciplinary tumor board meeting during a dedicated consultation.
The objectives of external beam radiotherapy treatment for liver tumors are to destroy cancer cells and reduce the tumor’s size. There are two main types of external beam radiotherapy: conformal radiotherapy and stereotactic radiotherapy, which is currently the most widely used and recommended. This new technique allows for increased precision in delivering the radiation dose, reducing the number of sessions while maintaining its effectiveness. It uses 3D images to precisely locate the tumor and deliver a high dose of radiation directly to the tumor. This technique helps reduce the volume of irradiated healthy tissue, thereby minimizing side effects and improving treatment efficacy.
The treatment approach depends on the stage of the disease and the size of the tumor. External beam radiotherapy is usually administered in multiple sessions over a period of several weeks. During each session, patients lie on a treatment table, and a radiotherapy device is used to deliver a precise dose of radiation to the tumor. Anesthesia is not required for this procedure.
Note that this technique is not indicated for patients with:
- Unpreserved liver function;
- Extrahepatic metastases;
Side effects of radiotherapy treatment may include fatigue, nausea, vomiting, loss of appetite, and abdominal pain. These side effects are generally temporary and can be managed with medications or symptomatic treatments.
Post-treatment surveillance is important to detect any recurrence or reappearance of the tumor. Patients will be regularly followed by their referring physician, and imaging examinations such as CT scans or MRIs will be conducted every 3 months to monitor the disease’s progression. The true effectiveness of the treatment is typically assessed around 3 to 6 months after treatment due to the prolonged action of radiation.
In conclusion, external beam radiotherapy is a treatment option for patients with primarily single liver tumors that are not accessible to local-regional treatments. Advanced techniques like stereotactic radiotherapy enable increased precision in delivering radiation doses and a reduction in side effects.
Is chemotherapy indicated in patients with hepatocellular carcinoma?
Advanced hepatocellular carcinoma (HCC) is a form of liver cancer that has spread to other parts of the body. In addition to local-regional treatments like surgery or ablation, systemic treatment is often used to target cancer cells throughout the body. Three commonly used therapeutic approaches are Durvalumab-Tremelimumab, the combination of Atezolizumab-Bevacizumab, and tyrosine kinase inhibitors (TKIs).
Durvalumab-Tremelimumab is a combination of immunotherapy drugs used to stimulate the body’s immune system to recognize and fight advanced HCC cancer cells. These drugs target immune checkpoints to prevent cancer cells from evading the immune system. They can be administered intravenously at regular intervals as per your medical team’s recommendations.
The Atezolizumab-Bevacizumab combination is another therapeutic option used to treat advanced HCC. Atezolizumab is an immune checkpoint inhibitor, while Bevacizumab is an antibody that targets and inhibits the VEGF protein, which promotes the growth of blood vessels that feed tumors. This drug combination has shown significant improvement in overall survival for patients with advanced HCC.
Tyrosine kinase inhibitors (TKIs) are medications that block the growth signals of cancer cells by targeting specific enzymes involved in tumor growth. Drugs like sorafenib, lenvatinib, or other TKIs can be used to treat advanced HCC when other options are not suitable. These drugs are typically taken orally and require regular administration according to your medical team’s instructions.
It is important to note that each patient is unique, and the choice of treatment depends on various factors such as cancer stage, overall health status, and medical history. Your medical team will provide detailed information about the recommended treatment, including possible side effects.
Systemic treatments for advanced HCC can lead to side effects that vary from person to person. Common side effects include fatigue, nausea, vomiting, loss of appetite, liver problems, digestive issues, skin rashes, and changes in thyroid function. These side effects are closely monitored by your medical team, and appropriate measures will be taken to manage them and improve your quality of life during treatment.
In addition to side effects, regular monitoring is crucial to assess the treatment’s effectiveness and detect any new developments in the disease. Your medical team will recommend imaging assessments, such as liver scans, every 3 months or according to a schedule tailored to your individual situation. These imaging assessments help track the cancer’s progression and adjust the treatment if necessary.
Is chemotherapy indicated in patients with cholangiocarcinoma?
In cases where an individual has a liver tumor that cannot be safely removed through surgery or loco-regional therapies, or when the tumor has extended beyond the liver and affected other organs (metastasized), the most effective treatment option is chemotherapy. Chemotherapy is a medical intervention aimed at eliminating rapidly dividing cells, such as cancer cells, which grow at a faster rate than most other cells in the body, with the exception of blood cells. Until recently, the only systemic therapy that had demonstrated a survival benefit was the combination of cisplatin and gemcitabine. Both of these agents are generally well-tolerated and are administered in a three-week cycle. However, recent positive outcomes have led to the addition of immunotherapy (Durvalumab) to the cisplatin and gemcitabine combination, making it the new standard of care. Before each treatment cycle, a blood test is conducted to ensure that blood counts are at an acceptable level for the next round of chemotherapy. Common side effects include fatigue, nausea, vomiting, loss of appetite, liver issues, digestive problems, skin rashes, and changes in thyroid function. These side effects are closely monitored by your medical team, who will take appropriate measures to manage them and enhance your quality of life during treatment.
Apart from managing side effects, it is crucial to regularly monitor the treatment’s effectiveness and identify any new developments in the disease. Your medical team will recommend periodic imaging assessments, such as liver scans, every three months or according to a personalized schedule based on your specific condition. These imaging assessments help track the progression of cancer and allow for adjustments to the treatment plan if needed.
In cases where the disease continues to progress despite the initial therapy, alternative chemotherapy agents are employed as “second-line” treatments, which means they are used after the standard therapy has proven ineffective. A recent development in this regard is the use of folinic acid (also known as leucovorin or calcium folinate), fluorouracil (5FU), and oxaliplatin (FOLFOX) in second-line therapy, following tumor growth despite previous treatment. FOLFOX is administered every 14 days. Subsequently, 5FU is administered through an infusion pump for 46 hours. This delivery method is similar to how some diabetics receive insulin, with a needle inserted under the skin and a tube connected to a wearable pump for medication delivery. After the infusion is completed, there is a 12-day rest period with no treatment. This completes one cycle of FOLFOX. The primary adverse events associated with FOLFOX include fatigue, low blood counts, peripheral neuropathy (numbness and tingling in the extremities), and nausea and vomiting.
The approach to systemic therapy in cholangiocarcinoma has evolved significantly in recent years. This type of tumor is highly heterogeneous, meaning that different tumors may have distinct genetic characteristics. A novel approach involves analyzing the genetic alterations in the tumor and tailoring treatment based on these specific changes, a concept known as personalized medicine. For example, a drug called ivosedinib has demonstrated promising results for cancers carrying a specific mutation known as IDH1. Furthermore, numerous clinical trials are underway to evaluate the effectiveness of potential drugs in patients with abnormalities in a gene called FGFR2.
What is the role of caregivers and family?
Individuals undergoing cancer treatment require substantial physical and emotional support, which can come from family members, friends, or a hired caregiver. Chemotherapy can be physically taxing, leading to fatigue, nausea, vomiting, and a need for special precautions to prevent infections. Additionally, there is the emotional burden of dealing with the effects of cancer and its treatment, as well as concerns about the possibility of mortality. All of these aspects necessitate not only emotional support but also practical assistance, such as preparing appetizing meals and, for bedridden patients, attending to toileting needs.
For patients who are mobile, getting to and from the treatment center can be challenging. Post-treatment symptoms may limit their ability to drive or use public transportation, making it the responsibility of the caregiver to provide transportation. Patients may also struggle with grocery shopping, which may require the caregiver’s assistance. These responsibilities can place a significant strain on family members, and it is advisable to distribute the workload among family members or, when feasible, enlist the help of a professional caregiver.
Disclaimer:
Information at www.ilca-online.org is general information provided for educational or reference purposes only and is not a substitute for professional care. It is continually being updated and so may not be accurate, current or complete and is subject to change without notice. No information obtained on this site should be relied on as the basis for treating or diagnosing conditions, symptoms, or illness and all queries should be directed to your health professional.
Acknowledgement
@Lili Gundelach – DiCE Project Coordinator in charge of Liver Cancer
@Ruel Jacob – DiCE Communication Manager
@Luca Arfini – DICE Communication Coordinator
